2022
Laurin D; Mercier C; Quansah N; Robert J S; Usson Y; Schneider D; Hindré T; Schaack B
Extracellular vesicles from 50,000 generation clones of the Escherichia coli long-term evolution experiment Journal Article
International Journal of Molecular Sciences, 23 , pp. 14580, 2022, ISSN: 1422-0067.
Abstract | Links | BibTeX | Altmetric | Tags: Cell Morphology, Genotypes and Phenotypes
@article{Laurin2022,
title = {Extracellular vesicles from 50,000 generation clones of the \textit{Escherichia coli} long-term evolution experiment},
author = {David Laurin and Corinne Mercier and Nyamekye Quansah and Julie Suzanne Robert and Yves Usson and Dominique Schneider and Thomas Hindré and Béatrice Schaack },
url = {https://www.mdpi.com/1422-0067/23/23/14580},
doi = {10.3390/ijms232314580},
issn = {1422-0067},
year = {2022},
date = {2022-11-01},
urldate = {2022-11-01},
journal = {International Journal of Molecular Sciences},
volume = {23},
pages = {14580},
abstract = {Extracellular vesicles (EVs) are critical elements of cell–cell communication. Here, we characterized the outer membrane vesicles (OMVs) released by specific clones of Escherichia coli isolated from the Long-Term Evolution Experiment after 50,000 generations (50K) of adaptation to glucose minimal medium. Compared with their ancestor, the evolved clones produce small OMVs but also larger ones which display variable amounts of both OmpA and LPS. Tracking ancestral, fluorescently labelled OMVs revealed that they fuse with both ancestral- and 50K-evolved cells, albeit in different proportions. We quantified that less than 2% of the cells from one 50K-evolved clone acquired the fluorescence delivered by OMVs from the ancestral strain but that one cell concomitantly fuses with several OMVs. Globally, our results showed that OMV production in E. coli is a phenotype that varies along bacterial evolution and question the contribution of OMVs-mediated interactions in bacterial adaptation.},
keywords = {Cell Morphology, Genotypes and Phenotypes},
pubstate = {published},
tppubtype = {article}
}
Marshall D J; Malerba M; Lines T; Sezmis A L; Hasan C M; Lenski R E; McDonald M J
Long-term experimental evolution decouples size and production costs in Escherichia coli Journal Article
Proceedings of the National Academy of Sciences of the United States of America, 119 (21), pp. e2200713119, 2022.
Abstract | Links | BibTeX | Altmetric | Tags: Cell Morphology, Demography and Ecology
@article{marshall2021,
title = {Long-term experimental evolution decouples size and production costs in \emph{Escherichia coli}},
author = {Dustin J. Marshall and Martino Malerba and Thomas Lines and Aysha L. Sezmis and Chowdhury M. Hasan and Richard E. Lenski and Michael J. McDonald},
editor = {Ruth Shaw},
url = {https://pnas.org/doi/full/10.1073/pnas.2200713119},
doi = {10.1073/pnas.2200713119},
year = {2022},
date = {2022-05-20},
urldate = {2022-05-20},
journal = {Proceedings of the National Academy of Sciences of the United States of America},
volume = {119},
number = {21},
pages = {e2200713119},
abstract = {Body size covaries with population dynamics across life’s domains. Metabolism may impose fundamental constraints on the coevolution of size and demography, but experimental tests of the causal links remain elusive. We leverage a 60,000-generation experiment in which \textit{Escherichia coli} populations evolved larger cells to examine intraspecific metabolic scaling and correlations with demographic parameters. Over the course of their evolution, the cells have roughly doubled in size relative to their ancestors. These larger cells have metabolic rates that are absolutely higher, but relative to their size, they are lower. Metabolic theory successfully predicted the relations between size, metabolism, and maximum population density, including support for Damuth’s law of energy equivalence, such that populations of larger cells achieved lower maximum densities but higher maximum biomasses than populations of smaller cells. The scaling of metabolism with cell size thus predicted the scaling of size with maximum population density. In stark contrast to standard theory, however, populations of larger cells grew faster than those of smaller cells, contradicting the fundamental and intuitive assumption that the costs of building new individuals should scale directly with their size. The finding that the costs of production can be decoupled from size necessitates a reevaluation of the evolutionary drivers and ecological consequences of biological size more generally.},
howpublished = {bioRxiv},
keywords = {Cell Morphology, Demography and Ecology},
pubstate = {published},
tppubtype = {article}
}
2021
van Raay K; Stolyar S; Sevigny J; Draghi J; Lenski R E; Marx C J; Kerr B; Zaman L
Evolution with private resources reverses some changes from long-term evolution with public resources Unpublished
bioRxiv, 2021.
Abstract | Links | BibTeX | Altmetric | Tags: Cell Morphology, Correlated Responses, Demography and Ecology, Descendant Experiments, Genotypes and Phenotypes, Historical Contingency, Methods and Miscellaneous
@unpublished{Raay2021,
title = {Evolution with private resources reverses some changes from long-term evolution with public resources},
author = {Katrina {van Raay} and Sergey Stolyar and Jordana Sevigny and Jeremy Draghi and Richard E. Lenski and Christopher J. Marx and Benjamin Kerr and Luis Zaman},
url = {https://www.biorxiv.org/content/10.1101/2021.07.11.451942v1},
doi = {https://doi.org/10.1101/2021.07.11.451942},
year = {2021},
date = {2021-07-12},
urldate = {2021-07-12},
journal = {bioRxiv},
pages = {2021.07.11.451942},
abstract = {A population under selection to improve one trait may evolve a sub-optimal state for another trait due to tradeoffs and other evolutionary constraints. How this evolution affects the capacity of a population to adapt when conditions change to favor the second trait is an open question. We investigated this question using isolates from a lineage spanning 60,000 generations of the Long-Term Evolution Experiment (LTEE) with \textit{Escherichia coli}, where cells have access to a shared pool of resources, and have evolved increased competitive ability and a concomitant reduction in numerical yield. Using media-in oil emulsions we shifted the focus of selection to numerical yield, where cells grew in isolated patches with private resources. We found that the time spent evolving under shared resources did not affect the ability to re-evolve toward higher numerical yield. The evolution of numerical yield commonly occurred through mutations in the phosphoenolpyruvate phosphotransferase system. These mutants exhibit slower uptake of glucose, making them poorer competitors for public resources, and produce smaller cells that release less carbon as overflow metabolites. Our results demonstrate that mutations that were not part of adaptation under one selective regime may enable access to ancestral phenotypes when selection changes to favor evolutionary reversion. },
howpublished = {bioRxiv},
keywords = {Cell Morphology, Correlated Responses, Demography and Ecology, Descendant Experiments, Genotypes and Phenotypes, Historical Contingency, Methods and Miscellaneous},
pubstate = {published},
tppubtype = {unpublished}
}
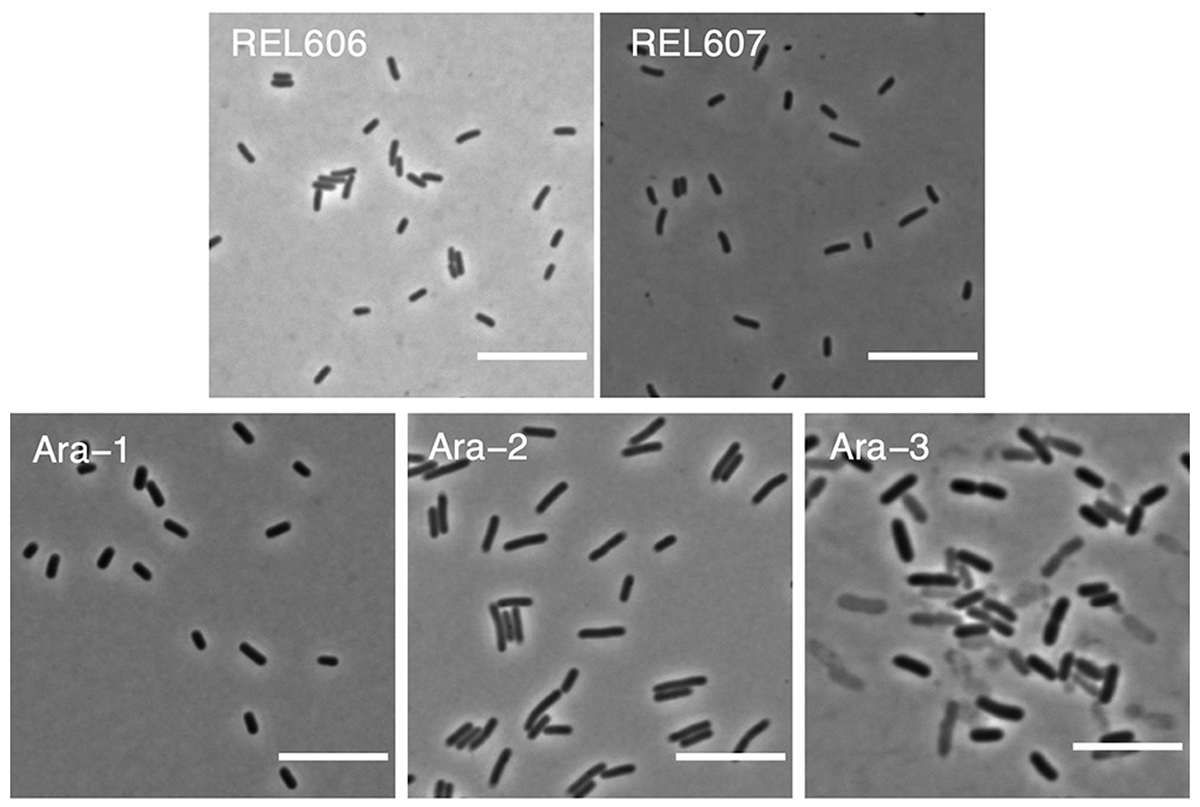
Grant N A; abdel Magid A; Franklin J; Dufour Y; Lenski R E
Changes in cell size and shape during 50,000 generations of experimental evolution with Escherichia coli Journal Article
Journal of Bacteriology, 203 (10), pp. e00469-20, 2021.
Abstract | Links | BibTeX | Altmetric | Tags: Cell Morphology, Genotypes and Phenotypes
@article{Grant2021,
title = {Changes in cell size and shape during 50,000 generations of experimental evolution with \textit{Escherichia coli}},
author = {Nkrumah A. Grant and Ali {abdel Magid} and Joshua Franklin and Yann Dufour and Richard E. Lenski},
url = {https://journals.asm.org/doi/10.1128/JB.00469-20},
doi = {10.1128/JB.00469-20},
year = {2021},
date = {2021-01-01},
urldate = {2021-01-01},
journal = {Journal of Bacteriology},
volume = {203},
number = {10},
pages = {e00469-20},
abstract = {Bacteria adopt a wide variety of sizes and shapes, with many species exhibiting stereotypical morphologies. How morphology changes, and over what timescales, is less clear. Previous work examining cell morphology in an experiment with \textit{Escherichia coli} showed that populations evolved larger cells and, in some cases, cells that were less rod-like. That experiment has now run for over two more decades. Meanwhile, genome sequence data are available for these populations, and new computational methods enable high-throughput microscopic analyses. In this study, we measured stationary-phase cell volumes for the ancestor and 12 populations at 2,000, 10,000, and 50,000 generations, including measurements during exponential growth at the last time point. We measured the distribution of cell volumes for each sample using a Coulter counter and microscopy, the latter of which also provided data on cell shape. Our data confirm the trend toward larger cells while also revealing substantial variation in size and shape across replicate populations. Most populations first evolved wider cells but later reverted to the ancestral length-to-width ratio. All but one population evolved mutations in rod shape maintenance genes. We also observed many ghost-like cells in the only population that evolved the novel ability to grow on citrate, supporting the hypothesis that this lineage struggles with maintaining balanced growth. Lastly, we show that cell size and fitness remain correlated across 50,000 generations. Our results suggest that larger cells are beneficial in the experimental environment, while the reversion toward ancestral length-to-width ratios suggests partial compensation for the less favorable surface area-to-volume ratios of the evolved cells.},
keywords = {Cell Morphology, Genotypes and Phenotypes},
pubstate = {published},
tppubtype = {article}
}
2009
Stanek M T; Cooper T F; Lenski R E
Identification and dynamics of a beneficial mutation in a long-term evolution experiment with Escherichia coli Journal Article
BMC Evolutionary Biology, 9 (1), pp. 302, 2009, ISSN: 1471-2148.
Abstract | Links | BibTeX | Altmetric | Tags: Cell Morphology, Genome Evolution
@article{Stanek2009,
title = {Identification and dynamics of a beneficial mutation in a long-term evolution experiment with \textit{Escherichia coli}},
author = {Mark T. Stanek and Tim F. Cooper and Richard E. Lenski},
url = {https://bmcevolbiol.biomedcentral.com/articles/10.1186/1471-2148-9-302},
doi = {10.1186/1471-2148-9-302},
issn = {1471-2148},
year = {2009},
date = {2009-12-01},
urldate = {2009-12-01},
journal = {BMC Evolutionary Biology},
volume = {9},
number = {1},
pages = {302},
abstract = {Background
Twelve populations of \textit{E. coli} were serially propagated for 20,000 generations in a glucose-supplemented minimal medium in order to study the dynamics of evolution. We sought to find and characterize one of the beneficial mutations responsible for the adaptation and other phenotypic changes, including increased cell size, in one of these populations.
Results
We used transposon-tagging followed by P1-transduction into the ancestor, screening for increased cell size and fitness, co-transduction analysis, and DNA sequencing. We identified a 1-bp insertion in the BoxG1 region located upstream of \textit{glmUS}, an operon involved in cell-wall biosynthesis. When transduced into the ancestor, this mutation increased competitive fitness by about 5%. This mutation spread through its population of origin between 500 and 1500 generations. Mutations in this region were not found in the other 11 evolving populations, even after 20,000 generations.
Conclusion
The 1-bp insertion in the BoxG1 region near \textit{glmUS} was demonstrably beneficial in the environment in which it arose. The absence of similar mutations in the other evolved populations suggests that they substituted other mutations that rendered this particular mutation unimportant. These results show the unpredictability of adaptive evolution, whereas parallel substitutions at other loci in these same populations reveal the predictability. },
keywords = {Cell Morphology, Genome Evolution},
pubstate = {published},
tppubtype = {article}
}
Twelve populations of E. coli were serially propagated for 20,000 generations in a glucose-supplemented minimal medium in order to study the dynamics of evolution. We sought to find and characterize one of the beneficial mutations responsible for the adaptation and other phenotypic changes, including increased cell size, in one of these populations.
Results
We used transposon-tagging followed by P1-transduction into the ancestor, screening for increased cell size and fitness, co-transduction analysis, and DNA sequencing. We identified a 1-bp insertion in the BoxG1 region located upstream of glmUS, an operon involved in cell-wall biosynthesis. When transduced into the ancestor, this mutation increased competitive fitness by about 5%. This mutation spread through its population of origin between 500 and 1500 generations. Mutations in this region were not found in the other 11 evolving populations, even after 20,000 generations.
Conclusion
The 1-bp insertion in the BoxG1 region near glmUS was demonstrably beneficial in the environment in which it arose. The absence of similar mutations in the other evolved populations suggests that they substituted other mutations that rendered this particular mutation unimportant. These results show the unpredictability of adaptive evolution, whereas parallel substitutions at other loci in these same populations reveal the predictability.
Philippe N; Pelosi L; Lenski R E; Schneider D
Evolution of Penicillin-Binding Protein 2 Concentration and Cell Shape during a Long-Term Experiment with Escherichia coli Journal Article
Journal of Bacteriology, 191 (3), pp. 909-921, 2009, ISSN: 0021-9193.
Abstract | Links | BibTeX | Altmetric | Tags: Cell Morphology, Genotypes and Phenotypes
@article{nokey,
title = {Evolution of Penicillin-Binding Protein 2 Concentration and Cell Shape during a Long-Term Experiment with \textit{Escherichia coli}},
author = {Nadège Philippe and Ludovic Pelosi and Richard E. Lenski and Dominique Schneider
},
url = {https://journals.asm.org/doi/full/10.1128/JB.01419-08},
doi = {10.1128/JB.01419-08},
issn = {0021-9193},
year = {2009},
date = {2009-02-01},
urldate = {2009-02-01},
journal = {Journal of Bacteriology},
volume = {191},
number = {3},
pages = {909-921},
abstract = {Peptidoglycan is the major component of the bacterial cell wall and is involved in osmotic protection and in determining cell shape. Cell shape potentially influences many processes, including nutrient uptake as well as cell survival and growth. Peptidoglycan is a dynamic structure that changes during the growth cycle. Penicillin-binding proteins (PBPs) catalyze the final stages of peptidoglycan synthesis. Although PBPs are biochemically and physiologically well characterized, their broader effects, especially their effects on organismal fitness, are not well understood. In a long-term experiment, 12 populations of \textit{Escherichia coli} having a common ancestor were allowed to evolve for more than 40,000 generations in a defined environment. We previously identified mutations in the \textit{pbpA} operon in one-half of these populations; this operon encodes PBP2 and RodA proteins that are involved in cell wall elongation. In this study, we characterized the effects of two of these mutations on competitive fitness and other phenotypes. By constructing and performing competition experiments with strains that are isogenic except for the \textit{pbpA} alleles, we showed that both mutations that evolved were beneficial in the environment used for the long-term experiment and that these mutations caused parallel phenotypic changes. In particular, they reduced the cellular concentration of PBP2, thereby generating spherical cells with an increased volume. In contrast to their fitness-enhancing effect in the environment where they evolved, both mutations decreased cellular resistance to osmotic stress. Moreover, one mutation reduced fitness during prolonged stationary phase. Therefore, alteration of the PBP2 concentration contributed to physiological trade-offs and ecological specialization during experimental evolution.},
keywords = {Cell Morphology, Genotypes and Phenotypes},
pubstate = {published},
tppubtype = {article}
}
2000
Lenski R E; Mongold J A
Cell Size, Shape, and Fitness in Evolving Populations of Bacteria Book Chapter
Scaling in Biology, Oxford University Press, Inc., USA, 2000, ISBN: 019513141X.
Abstract | BibTeX | Tags: Cell Morphology, Correlated Responses, Demography and Ecology
@inbook{10.5555/345504.345526,
title = {Cell Size, Shape, and Fitness in Evolving Populations of Bacteria},
author = {Richard E. Lenski and Judith A. Mongold},
isbn = {019513141X},
year = {2000},
date = {2000-01-01},
urldate = {2000-01-01},
booktitle = {Scaling in Biology},
publisher = {Oxford University Press, Inc.},
address = {USA},
abstract = {There is a long and substantial history of studying allometric scaling relationships in animals and plants, which is well represented by other chapters in this volume. These studies have relevance for many fields, from cardiovascular physiology to community ecology. From the perspective of evolutionary biology, scaling relationships are important because they provide an empirical focus for investigating the tensions between structural constraints, on the one hand, and natural selection, on the other, as they vie to shape—quite literally—organisms and life histories. Despite the breadth of research on allometric scaling, those organisms at the microscopic end of the scale, especially bacteria, have been largely ignored.},
keywords = {Cell Morphology, Correlated Responses, Demography and Ecology},
pubstate = {published},
tppubtype = {inbook}
}
1996
Mongold J A; Lenski R E
Experimental rejection of a nonadaptive explanation for increased cell size in Escherichia coli Journal Article
Journal of Bacteriology, 178 (17), pp. 5333–5334, 1996, ISSN: 0021-9193.
Abstract | Links | BibTeX | Altmetric | Tags: Cell Morphology
@article{Mongold1996,
title = {Experimental rejection of a nonadaptive explanation for increased cell size in \textit{Escherichia coli}},
author = {Judith A. Mongold and Richard E. Lenski},
url = {https://journals.asm.org/doi/10.1128/jb.178.17.5333-5334.1996},
doi = {10.1128/jb.178.17.5333-5334.1996},
issn = {0021-9193},
year = {1996},
date = {1996-09-01},
urldate = {1996-09-01},
journal = {Journal of Bacteriology},
volume = {178},
number = {17},
pages = {5333--5334},
abstract = {Populations of \textit{Escherichia coli} that have been serially propagated for thousands of generations in glucose minimal medium show heritable increases in both cell size and growth rate. We sought to test the hypothesis that the increased cell size of the derived genotypes could be explained solely by their faster growth. The regression of cell size on growth rate differed significantly between populations having ancestral and derived genotypes, with the latter producing larger cells over almost the entire range of growth rates. Thus, the physiological coupling between cell size and growth rate has been evolutionarily altered.},
keywords = {Cell Morphology},
pubstate = {published},
tppubtype = {article}
}
Elena S F; Cooper V S; Lenski R E
Punctuated Evolution Caused by Selection of Rare Beneficial Mutations Journal Article
Science, 272 (5269), pp. 1802–1804, 1996, ISSN: 0036-8075.
Abstract | Links | BibTeX | Altmetric | Tags: Cell Morphology
@article{Elena1996,
title = {Punctuated Evolution Caused by Selection of Rare Beneficial Mutations},
author = {Santiago F. Elena and Vaughn S. Cooper and Richard E. Lenski},
url = {https://www.sciencemag.org/lookup/doi/10.1126/science.272.5269.1802},
doi = {10.1126/science.272.5269.1802},
issn = {0036-8075},
year = {1996},
date = {1996-06-01},
urldate = {1996-06-01},
journal = {Science},
volume = {272},
number = {5269},
pages = {1802--1804},
abstract = {For more than two decades there has been intense debate over the hypothesis that most morphological evolution occurs during relatively brief episodes of rapid change that punctuate much longer periods of stasis. A clear and unambiguous case of punctuated evolution is presented for cell size in a population of \textit{Escherichia coli} evolving for 3000 generations in a constant environment. The punctuation is caused by natural selection as rare, beneficial mutations sweep successively through the population. This experiment shows that the most elementary processes in population genetics can give rise to punctuated evolutionary dynamics.},
keywords = {Cell Morphology},
pubstate = {published},
tppubtype = {article}
}
1995
Travisano M; Mongold J A; Bennett A F; Lenski R E
Experimental Tests of the Roles of Adaptation, Chance, and History Journal Article
Science, 267 (January), 1995.
Abstract | Links | BibTeX | Altmetric | Tags: Cell Morphology, Correlated Responses, Descendant Experiments, Fitness Trajectories, Historical Contingency, Methods and Miscellaneous, Parallelism and Divergence
@article{Travisano1995,
title = {Experimental Tests of the Roles of Adaptation, Chance, and History},
author = {Michael Travisano and Judith A. Mongold and Albert F. Bennett and Richard E. Lenski},
url = {https://www.science.org/lookup/doi/10.1126/science.7809610},
doi = {https://doi.org/10.1126/science.7809610},
year = {1995},
date = {1995-01-01},
urldate = {1995-01-01},
journal = {Science},
volume = {267},
number = {January},
abstract = {The contributions of adaptation, chance, and history to the evolution of fitness and cell size were measured in two separate experiments using bacteria. In both experiments, populations propagated in identical environments achieved similar fitnesses, regardless of prior history or subsequent chance events. In contrast, the evolution of cell size, a trait weakly correlated with fitness, was more strongly influenced by history and chance.},
keywords = {Cell Morphology, Correlated Responses, Descendant Experiments, Fitness Trajectories, Historical Contingency, Methods and Miscellaneous, Parallelism and Divergence},
pubstate = {published},
tppubtype = {article}
}
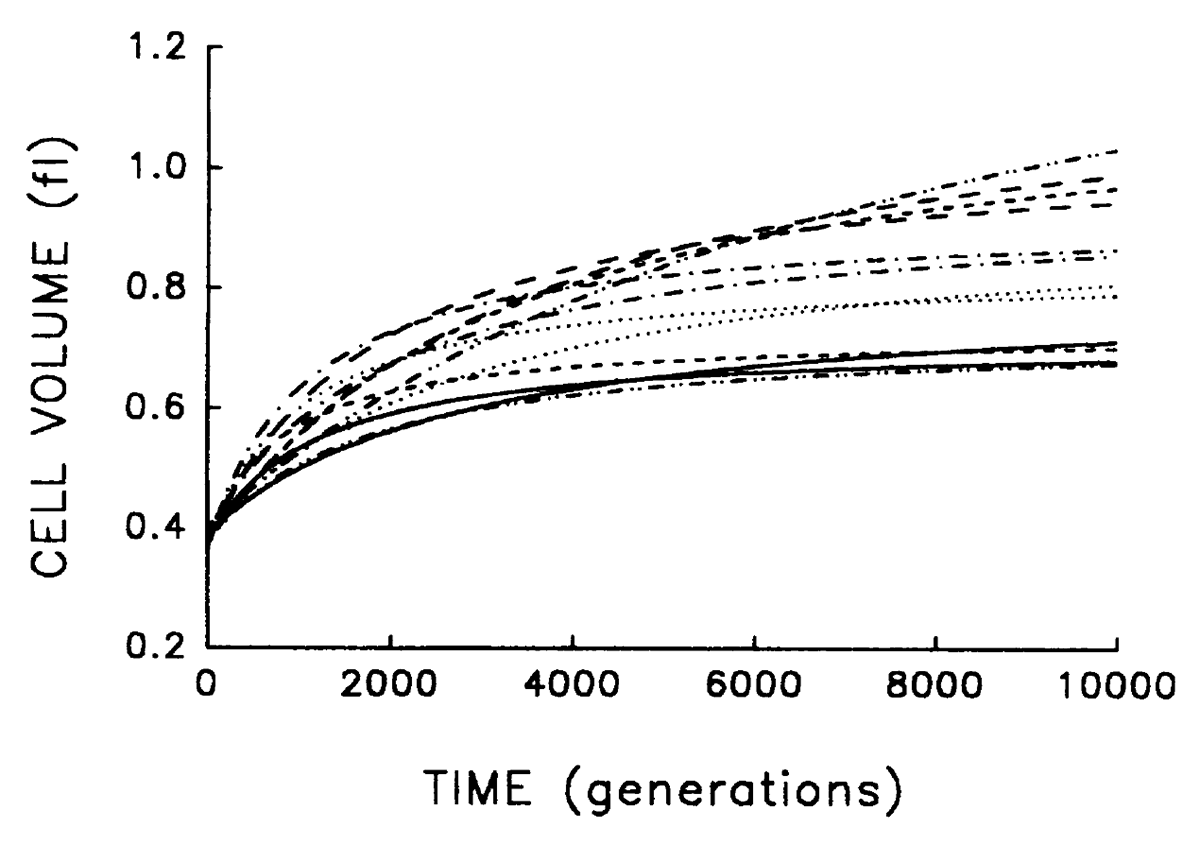
1994
Lenski R E; Travisano M
Dynamics of adaptation and diversification: a 10,000-generation experiment with bacterial populations. Journal Article
Proceedings of the National Academy of Sciences of the United States of America, 91 (15), pp. 6808–6814, 1994, ISSN: 0027-8424.
Abstract | Links | BibTeX | Altmetric | Tags: Cell Morphology, Fitness Trajectories, Parallelism and Divergence
@article{Lenski1994,
title = {Dynamics of adaptation and diversification: a 10,000-generation experiment with bacterial populations.},
author = {Richard E. Lenski and Michael Travisano},
url = {http://www.pnas.org/cgi/doi/10.1073/pnas.91.15.6808},
doi = {10.1073/pnas.91.15.6808},
issn = {0027-8424},
year = {1994},
date = {1994-07-01},
urldate = {1994-07-01},
journal = {Proceedings of the National Academy of Sciences of the United States of America},
volume = {91},
number = {15},
pages = {6808--6814},
abstract = {We followed evolutionary change in 12 populations of \textit{Escherichia coli} propagated for 10,000 generations in identical environments. Both morphology (cell size) and fitness (measured in competition with the ancestor) evolved rapidly for the first 2000 generations or so after the populations were introduced into the experimental environment, but both were nearly static for the last 5000 generations. Although evolving in identical environments, the replicate populations diverged significantly from one another in both morphology and mean fitness. The divergence in mean fitness was sustained and implies that the populations have approached different fitness peaks of unequal height in the adaptive landscape. Although the experimental time scale and environment were microevolutionary in scope, our experiments were designed to address questions concerning the origin as well as the fate of genetic and phenotypic novelties, the repeatability of adaptation, the diversification of lineages, and thus the causes and consequences of the uniqueness of evolutionary history. In fact, we observed several hallmarks of macroevolutionary dynamics, including periods of rapid evolution and stasis, altered functional relationships between traits, and concordance of anagenetic and cladogenetic trends. Our results support a Wrightian interpretation, in which chance events (mutation and drift) play an important role in adaptive evolution, as do the complex genetic interactions that underlie the structure of organisms.},
keywords = {Cell Morphology, Fitness Trajectories, Parallelism and Divergence},
pubstate = {published},
tppubtype = {article}
}